A research team led by Shen Hanming, chair professor in the Faculty of Health Sciences (FHS) at the University of Macau (UM), has made a breakthrough in clarifying the mechanism by which SMAD Family Member 3 (SMAD3), a key effector in the transforming growth factor beta (TGFβ)-SMAD signalling pathway, regulates PTEN-induced kinase-1 (PINK1) transcription. This study has important theoretical value in elucidating the pathological function of PINK1 in neurodegenerative diseases such as Parkinson’s disease. The findings have been published in the internationally renowned journal Cell Discovery.
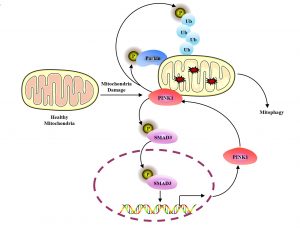
Mitophagy is a selective autophagy. As the core mechanism of mitochondrial quality control, it selectively removes damaged mitochondria through the autophagy-lysosome pathway to maintain mitochondrial homeostasis. The PINK1-Parkin pathway is an important regulatory mechanism of mitophagy. When mitochondria are damaged, the PINK1 protein becomes stabilised in the mitochondrial outer membrane and is activated by autophosphorylation to form a positive feedback loop by phosphorylating ubiquitin (Ub) and E3 ubiquitin ligase Parkin for continuously amplifying mitophagy signals. In recent years, there have been in-depth studies on the regulatory mechanism of PINK1-Parkin downstream, but the regulation of PINK1 itself under mitochondrial damage, especially for its transcriptional regulation, remains unclear.
Previous studies have shown that PINK1 stability induced by mitochondrial damage mainly depends on its inability to import into mitochondrial inner membrane, thereby escaping the cleavage by MMP and PARL proteases, ultimately accumulating in mitochondrial outer membrane. This mechanism is considered to be the main reason for the accumulation of PINK1 under mitochondrial damage conditions. Prof Shen’s research team found that the transcriptional regulation of PINK1 after mitochondrial damage is also a key reason for the accumulation of PINK1. They identified SMAD3 as a potential transcription factor for PINK1 through bioinformatics analysis. Further experimental verification showed that mitochondrial depolarisation can significantly promote the phosphorylation and nuclear translocation of SMAD3, thereby upregulating the transcription levels of PINK1. This regulatory process is independent of the key components in the classical TGFβ signalling pathway (such as TGFβ-R1, SMAD2, or SMAD4). Further studies have found that PINK1 activation induced by mitochondrial depolarisation is also involved in its own transcriptional regulation: PINK1, as a protein kinase of SMAD3, activates SMAD3 through phosphorylation, thereby promoting the transcriptional expression of PINK1, forming a positive feedback loop and accelerating the process of mitophagy.
Mitophagy is a key cell survival mechanism that prevents cell death caused by mitochondrial damage by clearing damaged mitochondria. The study revealed that SMAD3 establishes an anti-apoptotic signalling pathway by activating the transcription of PINK1, providing an important survival mechanism for cells in response to mitochondrial damage. The study also discovered a new function of SMAD3 in the transcriptional regulation of PINK1, revealed the mutual positive feedback regulatory mechanism between PINK1 and SMAD3, expanded the understanding of the non-classical biological functions of SMAD3, and clarified its new functions in mitochondrial quality control and cell fate regulation. This mutual regulatory mechanism improves the efficiency of mitophagy. The study provides a new research perspective for a deeper understanding of the fine regulatory mechanism of mitophagy and shows an important theoretical value for clarifying the pathological function of PINK1 in neurodegenerative diseases such as Parkinson’s disease.
The corresponding author of the study is Prof Shen. The first author is Tang Mingzhu, a postdoctoral fellow in FHS. Significant contributions were also made by FHS doctoral students Rong Dade, Gao Xiangzheng, and Tang Haimei, as well as members of the Biological Imaging and Stem Cell Core, and the Genomics, Bioinformatics and Single Cell Analysis Core in FHS. The research project was funded by the Science and Technology Development Fund of the Macao SAR (File No.: 0078/2020/A2, 0031/2021/A1, 0081/2022/AMJ, 0004/2021/AKP), and UM (File No.: CPG2023-00032-FHS, MYRG2020-00022-FHS). The full version of the research article is available at: https://www.nature.com/articles/s41421-025-00774-4.
| Source: Faculty of Health Sciences | |
| Media Contact Information: | |
| Communications Office, University of Macau | |
| Albee Lei | Tel: (853) 8822 8004 |
| Bell Leong | Tel: (853) 8822 8009 |
| Email: | prs.media@um.edu.mo |